Abstract
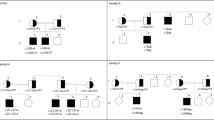
The persistent Müllerian duct syndrome (PMDS) is a rare form of male pseudohermaphroditism, characterized by the persistence of Müllerian derivatives in otherwise normal males. Two mutations, present in the homozygous state, have been previously described in such patients. The present observation is the first example of compound heterozygosity in this condition. DNA was obtained from a 3-month-old patient with PMDS, in whom no serum anti-Müllerian hormone (AMH) could be detected by enzyme-linked immunosorbent assay. Sequencing of cloned polymerase chain reaction amplified fragments of the AMH gene revealed a 14-bp deletion in the second exon of the maternal allele; this deletion disrupted the open reading frame. It occurred at a site containing two 8-bp direct repeats flanking a 6-bp sequence and removed one whole repeat plus all of the intervening sequence. It may be the result of a slipped mispairing at the DNA replication fork. The paternal allele contains a stop mutation in the third exon. These two mutations, impairing both AMH alleles, are consistent with the occurrence of PMDS, and are shared with a phenotypically normal younger sister. In this family, various other mutations, devoid of physiological significance, suggest that the AMH gene is highly polymorphic.
Similar content being viewed by others
References
Baker ML, Metcalfe SA, Hutson JM (1990) Serum levels of Müllerian inhibiting substance in boys from birth to 18 years, as determined by enzyme immunoassay. J Clin Endocrinol Metab 70:11–15
Bézard J, Vigier B, Tran D, Mauléon P, Josso N (1987) Immunocytochemical study of anti-Müllerian hormone in sheep ovarian follicles during fetal and postnatal development. J Reprod Fertil 80:509–516
Cate RL, Mattaliano RJ, Hession C, Tizard R, Farber NM, Cheung A, Ninfa EG, Frey AZ, Gash DJ, Chow EP, Fisher RA, Bertonis JM, Torres G, Wallner BP, Ramachandran KL, Ragin RC, Manganaro TF, MacLaughlin DT, Donahoe PK (1986) Isolation of the bovine and human genes for Müllerian inhibiting substance and expression of the human gene in animal cells. Cell 45:685–698
Cate RL, Donahoe PK, MacLaughlin DT (1990) Müllerian-inhibiting substance. In: Sporn MB, Roberts AB (eds) Handbook of experimental pharmacology, vol 95/II. Springer. Berlin Heidelberg NewYork, pp 179–210
Cohen-Haguenauer O, Picard JY, Mattei MG, Serero S, Nguyen VC, Tand MF de, Guerrier D, Hors-Cayla MC, Josso N, Frézal J (1987) Mapping of the gene for anti-Müllerian hormone to the short arm of human chromosome 19. Cytogenet Cell Genet 44:2–6
Efstratiadis A, Posakony JW, Maniatis T, Lawn RM, O'Connell C, Spritz RA, DeRiel JK, Forget BG, Weissman SM, Slightom JL, Blechl AE, Smithies O, Baralle FE, Shoulders CC, Proudfoot NJ (1980) The structure and evolution of the human β-globin gene family. Cell 21:653–668
Haqq C, Lee MM, Tizard R, Wysk M, DeMarinis J, Donahoe PK, Cate RL (1992) Isolation of the rat gene for Müllerian inhibiting substance. Genomics 12:665–669
Harbison MD, Magid MLS, Josso N, Mininberg DT, New MI (1991) Anti-Müllerian hormone in three intersex conditions. Ann Genet 34:226–232
Hudson PL, Douglas I, Donahoe PK, Cate RL, Epstein J, Pepinsky RB, MacLaughlin DT (1990) An immunoassay to detect human Müllerian inhibiting substance in males and females during normal development. J Clin Endocrinol Metab 70:16–22
Imbeaud S, Carré-Eusèbe D, Boussin L, Knebelmann B, Guerrier D, Josso N, Picard JY (1992) Biologie moléculaire de l'hormone anti-Müllérienne normale et pathologique. Ann Endocrinol 52:415–419
Josso N, Legeai L, Forest MG, Chaussain JL, Brauner R (1990) An enzyme-linked immunoassay for anti-Müllerian hormone: a new tool for the evaluation of testicular function in infants and children. J Clin Endocrinol Metab 70:23–27
Josso N, Boussin L, Knebelmann B, Nihoul-Fékété C, Picard JY (1991) Anti-Müllerian hormone and intersex states. Trends Endocrinol Metab 2:227–233
Knebelmann B, Boussin L, Guerrier D, Legeai L, Kahn A, Josso N, Picard JY (1991) Anti-Müllerian hormone Bruxelles: a non-sense mutation associated with the persistent Müllerian duct syndrome. Proc Natl Acad Sci USA 88:3767–3771
Krawczak M, Cooper DN (1991) Gene deletions causing human genetic disease: mechanisms of mutagenesis and the role of the local DNA environment. Hum Genet 86:425–441
Lemesle-Varloot L, Henrissat B, Gaboriaud C, Bissery V, Morgat A, Mornon JP (1990) Hydrophobic cluster analysis — procedures to derive structural and functional information from 2-D-representation of protein sequences. Biochimie 72:555–574
Münsterberg A, Lovell-Badge R (1991) Expression of the mouse anti-Müllerian hormone gene suggests a role in both male and female sex differentiation. Development 113:613–624
Nagamine CM, Chan K, Lau YFC (1989) A PCR artifact — generation of heteroduplexes. Am J Hum Genet 45:337–339
Orita M, Suzuki Y, Sekiya T, Hayashi K (1989) Rapid and sensitive detection of point mutations and DNA polymorphisms using the polymerase chain reaction. Genomics 5:874–879
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Takahashi M, Koide SS, Donahoe PK (1986) Müllerian inhibiting substance as oocvte meiosis inhibitor. Mol Cell Endocrinol 47:225–234
Ueno S, Manganaro TF, Donahoe PK (1988) Human recombinant Müllerian inhibiting substance inhibition of rat oocyte meiosis is reversed by epidermal growth factor in vitro. Endocrinology 123:1652–1659
Vigier B, Picard JY, Tran D, Legeai L, Josso N (1984) Production of anti-Müllerian hormone: another homology between Sertoli and granulosa cells. Endocrinology 114:1315–1320
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carré-Eusèbe, D., Imbeaud, S., Harbison, M. et al. Variants of the anti-Müllerian hormone gene in a compound heterozygote with the persistent Müllerian duct syndrome and his family. Hum Genet 90, 389–394 (1992). https://doi.org/10.1007/BF00220465
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00220465