Summary
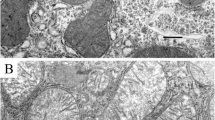
To assess the distribution of the thyroid hormone triiodothyronine (T3) within intact living cells, freshly prepared dispersed rat hepatocytes were incubated with [125I]-T3 for periods of 5 min and 30 min. Lightand electron-microscopic (EM) radioautography was carried out to determine the distribution of grains over the isolated cells. Both procedures showed the grains distributed almost entirely over the cytoplasmic matrix rather than the nucleus. Grain counts under the EM were compared with expectation based on established quantitative methods. Only the mitochondria showed obvious and statistically significant grain counts, whereas the nucleus failed to accumulate grains in excess of expectations by chance alone based on area. The findings support the existence of mitochondrial binding of T3, presumably a prerequisite for its action in direct stimulation of the mitochondria.
Similar content being viewed by others
References
Bar-Sella P, Stein O, Gross J (1973) Electron microscope radioautography of 125I-triiodothyronine in rat posterior pituitary and median eminence. Endocrinology 93:1410–1422
Berry MN, Friend DS (1969) High yield preparation of isolated rat liver parenchymal cells.A biochemical fine structural study. J Cell Biol 43:506–520
Cheng S-y, Maxfield FR, Robbins J, Willingham MC, Pastan IH (1980) Receptor mediated uptake of 3,3′, 5-triiodothyro-Lthyronine by cultured fibroblasts. Proc Natl Acad Sci USA. 77:3425–3429
Gershon MD, Nunez E A (1970) Histochemical and radioautographic studies of serotonin and parafollicular cells in the thyroid gland of the prehibernating bat. Endocrinology 86:160–166
Goglia F, Torresani J, Bugli P, Barletta A, Liverini G (1981) In vitro binding of T3 to rat liver mitochondria. Pflügers Arch 390:120–124
Hubbard AL, Stubenbrok H (1979) An electron microscope autoradiographic study of the carbohydrate recognition sites in rat liver. II. Intracellular fates of the 125I-ligands. J Cell Biol 83:65–81
Hubbard AL, Wilson G, Ashwell G, Stubenbrok H (1979) An electron microscope autoradiographic study of the carbohydrate recognition sites in rat liver. I. Distribution of 125I-ligands among the liver cell types. J Cell Biol 83:47–64
Kriz BM, Fong BB, Goldfine ID (1981) Quantitative electron microscope autoradiography of [125I]-triiodothyronine: evidence for multiple sites of hormone localization. Clin Res 29:506A
Maxfield FR, Willingham MC, Pastan I, Dragsten P, Cheng S-y (1981) Binding and mobility of cell surface receptors for 3,3′, 5-triiodo-L-thyronine. Science 211:63–64
Nunez EA, Gershon MD (1972) Synthesis and storage of serotonin by parafollicular (C) cells of the thyroid gland of active, prehibernating and hibernating bats. Endocrinology 90:1009–1024
Oppenheimer JH (1979) Thyroid hormone action at the cellular level. Science 203:971–979
Oppenheimer JH, Koerner D, Schwartz HL, Surks MI (1972) Specific nuclear triiodothyronine binding sites in rat liver and kidney. J Clin Endocrin Metab 35:330–333
Pliam NB, Goldfine ID (1977) High affinity thyroid hormone binding sites on purified rat liver plasma membranes. Biochem Biophys Res Commun 79:166–172
Rogers AW (1971) Recent developments in the use of autoradiographic techniques with electron microscopy. Phil Trans Roy Soc (London) B261:159–171
Salpeter MM, Bachmann L, Salpeter EE (1969) Resolution in electron microscope radioautography. J Cell Biol 41:1–32
Segal J, Ingbar SH (1979) Stimulation by triiodothyronine of the in vitro uptake of sugars by rat thymocytes. J Clin Invest 63:507–515
Segal J, Ingbar SH (1981) Studies of the mechanism by which 3,5, 31-triiodothyronine stimulates 2-deoxyglucose uptake in rat thymocytes in vitro. Role of calcium and adenosine 3′: 5′-monophosphate. J Clin Invest 68:103–110
Smith DM, Hitchcock KR (1982) Autoradiographic demonstration of thyroid hormone binding to intact liver cells in vivo. Endocrinology 111:1015–1016
Sterling K (1979) Thyroid hormone action at the cell level. New Eng J Med 300:117–123
Sterling K, Milch PO (1975) Thyroid hormone binding by a component of mitochondrial membrane. Proc Natl Acad Sci USA 72:3225–3229
Sterling K, Milch PO, Brenner MA, Lazarus JH (1977) Thyroid hormone action: the mitochondrial pathway. Science 172:996–999
Sterling K, Lazarus JH, Milch PO, Sakurada T, Brenner MA (1978) Mitochondrial thyroid hormone receptor: localization and physiological significance. Science 201:1126–1129
Sterling K, Brenner MA, Sakurada T (1980) Rapid effect of triiodothyronine on the mitochondrial pathway in rat liver in vivo. Science 210:340–343
Torresani J, Gharbi J, Lissitzky S (1979) Autoradiographic detection of triiodothyronine in nuclei from normal and hypothyroid rat liver. Biochem Biophys Res Commun 86:371–378
Williams MA (1969) The assessment of electron microscope autoradiographs. Adv Opt Elect Mic 3:219–272
Author information
Authors and Affiliations
Additional information
Supported in part by the VA medical Research Service, Grant PCM-8100280 from the National Science Foundation, Grant AM 19743 from the US Public Health Service, and a grant from the Kroc Foundation
Rights and permissions
About this article
Cite this article
Sterling, K., Campbell, G.A., Taliadouros, G.S. et al. Mitochondrial binding of triiodothyronine (T3). Cell Tissue Res. 236, 321–325 (1984). https://doi.org/10.1007/BF00214233
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00214233