Summary
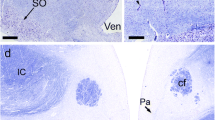
A vasopressin anti-idiotype antibody was generated by immunization with purified IgG of a primary vasopressin antiserum. The anti-idiotype antibody immunostained neurons in the supraoptic and paraventricular nuclei of the hypothalamus of normal and Brattleboro rats. The distribution of immunostained perikarya in these hypothalamic nuclei together with the staining of fibers in median eminence and neural lobe was similar to that observed in normal rats with anti-vasopressin and suggests strongly that vasopressinergic neurons are being stained. Absorption studies with vasopressin and a vasopressin-binding receptor protein further indicate that a receptor associated with vasopressinergic neurons is recognized by the anti-idiotype antibody.
Similar content being viewed by others
References
Amit T, Barkley RJ, Gavish M, Youdim MBH (1986) Anti-idiotype antibodies raised against anti-prolactin (PRL) antibodies recognize the PRL receptor. Endocrinology 118:835–843
Barkas T, Simpson JA (1982) Lack of inter-animal cross-reaction of anti-acetylcholine receptor antibodies at the receptor-binding site as demonstrated by heterologous anti-idiotype antisera: implications for immunotherapy of myasthenia gravis. Clin Exp Immunol 47:119–126
Baskin DG, Petracca F, Dorsa DM (1983) Autoradiographic localization of specific binding sites for [3H] (Arg8) vasopressin in the septum of the rat brain with tritium-sensitive film. Eur J Pharmacol 90:155–157
Buijs RM (1978) Intra- and extrahypothalamic vasopressin and oxytocin pathways in the rat.Pathways to the limbic system, medulla oblongata and spinal cord. Cell Tissue Res 192:423–435
Buijs RM (1983) Vasopressin and oxytocin — their role in neurotransmission. Pharmacol Ther Dest 22:127–141
Costantini MG and Pearlmutter AF (1984) Properties of the specific binding site for arginine vasopressin in rat hippocampal synaptic membranes. J Biol Chem 259:11739–11 745
Elias E, Maron R, Cohen IR, Shechter Y (1984) Mouse antibodies to the insulin receptor developing spontaneously as anti-idiotypes. Effects on glucose homeostasis and the insulin receptor. J Biol Chem 259:6416–6419
Guillet JG, Chamat S, Hoebeke J, Strosberg AD (1984) Production and detection of monoclonal anti-idiotype antibodies directed against a monoclonal anti-beta-adrenergic ligand antibody. J Immunol Methods 74:163–171
Jerne NK (1974) Towards a network theory of the immune system. Ann Immunol (Paris) 125 C:373–389
Junig JT, Abood LG, Skrobald AM (1986) Solubilization and purification of arginine vasopressin binding proteins from rat brain membranes. Peptides (in press)
Leng G, Wiersma J (1981) Effect of neural stalk stimulation on phasic discharge of supraoptic neurons in Brattleboro rats devoid of vasopressin. J Endocrinol 90:211–220
Luiten PGM, terHorst GJ, Karst H, Steffens AB (1985) The course of paraventricular hypothalamic efferents to autonomic structures in medulla and spinal cord. Brain Res 329:374–378
Melrose PA, Knigge KM (1985) Methods for the isolation of viable gonadotropin-releasing hormone (LRF) neurons. Peptides 6:347–351
Nisonoff A, Lamoyi E (1981) Implications of the presence of an internal image of the antigen in anti-idiotypic antibodies: Possible application to vaccine production. Clin Immunol Immunopathol 21:397–406
Pearlmutter AF, Costantini MG, Loeser B (1983) Characterization of (3H)-arginine vasopressin binding sites in paniculate preparations of rat brain. Peptides 4:335–341
Saper CB, Loewy AD, Swanson LW, Cowain WM (1976) Direct hypothalamo-autonomic connections. Brain Res 117:305–312
Shechter Y, Maron R, Elias D, Cohen IR (1982) Autoantibodies to insulin receptor spontaneously develop as anti-idiotypes in mice immunized with insulin. Science 216:542–545
Schmale H, Richter D (1984) Single base deletion in the vasopressin gene is the cause of diabetes insipidus in Brattleboro rats. Nature 308:705–709
Schreiber M, Fogelfeld L, Souroujon MC, Kohen F, Fuchs S (1983) Antibodies to spiroperidol and their anti-idiotypes as probes for studying dopamine receptors. Life Sci 33:1519–1526
Sofroniew MV, Weindl A (1978) Extrahypothalamic neurophysin-containing perikarya, fiber pathways and fiber clusters in the rat brain. Endocrinology 102:334–337
Swanson LW, Kuypers HGJM (1980) The paraventricular nucleus of the hypothalamus: cytoarchitectonic subdivisions and organization of projections to the pituitary, dorsal vagal complex, and spinal cord as demonstrated by retrograde fluorescent double-labeling methods. J Comp Neurol 194:555–570
Swanson LW, Sawchenko PE (1983) Hypothalamic integration: organization of the paraventricular and supraoptic nuclei. Ann Rev Neurosci 6:269–324
Van Leeuwen FW, Wolters P (1983) Light microscopic autoradiographic localization of (3H) arginine-vasopressin binding sites in the rat brain and kidney. Neurosci Lett 41:61–66
Walter R, Hoffman PL, Flexner JB, Flexner LB (1975) Neurohypophyseal hormones, analogs, and fragments: their effect on puromycin-induced amnesia. Proc Natl Acad Sci USA 72:4180–4184
Walter R, vanRee JM, DeWied E (1978) Modification of conditioned behavior of rats by neurohypophyseal hormones and analogues. Proc Natl Acad Sci USA 75:2493–2496
Wang BC, Share L, Crofton JT (1982) Central infusion of vasopressin decreased plasma concentrations in dogs. Am J Physiol 243:E365-E369
Yamamura HI, Gee KW, Brinton RE, Davis TP, Hadley M, Wamsley JK (1983) Light microscopic autoradiographic visualization of (3H)-arginine vasopressin binding sites in the rat brain. Life Sci 32:1919–1924
Author information
Authors and Affiliations
Additional information
Supported by NIH grants ES03239, NS18626 and NSF grant BNS-8310914. D.T.P. is the receipient of RCDA award NS00869
Rights and permissions
About this article
Cite this article
Knigge, K.M., Piekut, D.T. & Berlove, D.J. Immunocytochemistry of magnocellular neurons of supraoptic and paraventricular nuclei of normal and Brattleboro rats with vasopressin anti-idiotype antibody. Cell Tissue Res. 246, 509–513 (1986). https://doi.org/10.1007/BF00215190
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00215190