Summary
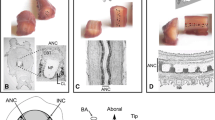
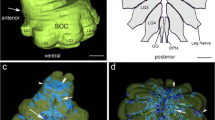
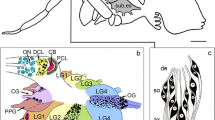
The distribution and ultrastructure of serotonin- and dopamine-immunoreactive (5-HTi and DAi) neurones have been investigated in the terminal ganglion of the cricket, Acheta domestica, using a pre-embedding chopper technique. Special attention has been paid to the immunoreactive structures in the neuropil. 5-HTi structures are extensively distributed and densely packed throughout the 5 neuromeres of the terminal ganglion and originate from several interneurones and efferent neurones. In contrast, DAi fibres are distributed sparsely although they extend to all neuromeres of the ganglion and originate from 6 interneurons only. For both 5-HTi and DAi neurones characteristic axonal projections and branching patterns can be distinguished. The 5-HTi axons exhibit rich varicose arborizations, whereas DAi neurones possess fewer varicosities in the neuropil. Electron microscopy shows that 5-HTi varicosities contain small (∼ 60 nm) and large (∼ 100 nm) agranular vesicles, and large (∼ 100 nm) granular vesicles, whereas in DAi varicosities small (∼ 60 nm) agranular and large (∼ 100 nm) granular vesicles are seen. Both 5-HTi and DAi varicosities form synaptic contacts. We conclude that both serotonin and dopamine may be used as neurotransmitters in the terminal ganglion of the cricket.
Similar content being viewed by others
References
Bishop CA, O'Shea M (1983) Serotonin immunoreactive neurons in the central nervous system of an insect (Periplaneta americana). J Neurobiol 14:251–269
Cook IM, Eraker J, Anderson JR (1969) The effect of various biogenic amines on the activity of the foregut of the cockroach, Blaberus giganteus. J Insect Physiol 15:445–455
Dambach M, Igelmund H (1983) Das Ei-Ablageverhalten von Grillen (Saltataria: Grylloida). Entomologia Generalis 8:267–281
David JC, Coulon JF (1985) Octopamine in invertebrates. A review. Prog Neurobiol 24:141–185
Davis NT (1985) Serotonin-immunoreactive visceral nerves and neurohemal system in the cockroach Periplaneta americana (L.). Cell Tissue Res 240:593–600
Dymond GR, Evans PD (1979) Biogenic amines in the nervous system of the cockroach, Periplaneta americana: Association of octopamine with mushroom bodies and dorsal unpaired median (DUM) neurones. Insect Biochem 9:535–545
Edwards JS (19177) One organism, several brains: Evolution and development of the insect central nervous system. Ann N Y Acad Sci 299:59–1
Edwards JS, Palka J (1974) The cerci and abdominal giant fibres of the house cricket Acheta domesticus. I. Anatomy and physiology of normal adults. Proc R Soc Lond [Biol] 185:83–103
Elekes K (1976) Electron microscope autoradiography of serotonin uptake in the ganglia of the fresh-water mussel, Anadonta cygnea L. Acta Biol Hung 27:183–189
Elekes K (1978) Autoradiographic localization of monoamine uptake in the central nervous system of a marine mollusc (Mactra stultorum L., Pelecypoda). Neuroscience 3:49–58
Elekes K (1979) Light and electron microscopic autoradiography of 3H-dopamine uptake in the ganglia of the freshwater mussel, Anodonta cygnea L. Acta Biol Acad Sci Hung 30:223–234
Evans PD (1980) Biogenic amines in the insect nervous system. Adv Insect Physiol 15:317–473
Flanagan TRJ (1983) Monoaminergic innervation in a hemipteran nervous system: a whole-mount histofluorescence survey. In: Strausfeld NJ (ed) Functional Neuroanatomy. Springer, Berlin Heidelberg New York Tokyo, pp 317–330
Geffard M, Buijs RM, Seguela P, Pool CW, LE Moal M (1984a) First demonstration of highly specific and sensitive antibodies against dopamine. Brain Res 294:161–165
Geffard M, Kah O, Onteniente B, Seguela P, Le Moal M, Delaage M (1984b) Antibodies to dopamine: Radioimmunological study of specificity in relation to immunocytochemistry. J Neurochem 42:1593–1599
Hiripi L, S-Rózsa K (1973) Fluorimetric determination of 5-hydroxytryptamine and catecholamines in the central nervous system and heart of Locusta migratoria migratorioides. J Insect Physiol 9:1481–1485
Hiripi L, S-Rózsa K (1981) Second messenger and monoamine receptors in the regulation of metamorphosis of Locusta migratoria migratorioides RF. In: S-Rózsa K (ed) Adv Physiol Sci, Vol 22, Neurotransmitters in invertebrates. Akadémiai Kiadó and Pergamon Press, Budapest Oxford pp 235–253
Howes EA, McLaughlin BJ, Heslop P (1974) The autoradiographical association of fast transport material with dense-core vesicles in the central nervous system of Anodonta cygnea L. Cell Tissue Res 153:545–558
Hustert R (1978) Segmental and interganglionic projections from primary fibres of insect mechanoreceptors. Cell Tissue Res 194:337–352
Hustert R, Topel U (1986) Location and major postembryonic changes of identified 5-HT-immunoreactivc neurons in the terminal ganglion of the cricket (Acheta domesticus). Cell Tissue Res 245:615–621
Kiss T, Varanka I, Benedeczky I (1984) Neuromuscular transmission in the visceral muscle of locust oviduct. Neuroscience 12:300–322
Klemm N (1976) Histochemistry of putative transmitter substances in the insect brain. Progr Neurobiol 7:99–169
Klemm N (1983) Detection of serotonin-containing neurons in the insect nervous system by antibodies to 5-HT. In: Strausfeld NJ (ed) Functional neuroanatomy. Springer Berlin Heidelberg New York Tokyo, pp 302–316
Klemm N, Hustert R (1984) The distribution of catecholamines and 5-HT in the stomatogastric nervous system of orthopteroid insects. In: Vizi ES, Magyar K (eds) Regulation of transmitter function. Académiai Kiadó, Budapest, pp 537–540
Klemm N, Steinbusch HWM, Sundler F (1984) Serotonin-immunoreactive neurons and their projections in the brain of the cockroach, Periplaneta americana. J Comp Neurol 225:387–395
Klemm N, Hustert R, Cantera R, Nässel DR (1986) Neurons reactive to antibodies against serotonin in the stomatogastric nervous system and in the alimentary canal of locust and crickets (Orthoptera, Insecta). Neuroscience 17:247–261
Livingstone MS, Schaeffer SF, Kravitz EA (1981) Biochemistry and ultrastructure of serotonergic nerve endings in the lobster: serotonin and octopamine are contained in different nerve endings. J Neurobiol 12:27–54
Lutz EM, Tyrer NM, Altman JS, Turner J (1985) Some insect sensory neurons contain 5-hydroxytryptamin. Brain Res 325:353–356
Mercer AR (1982) The effects of amines on behaviour and neuronal activity in the honey bee. In: Breed MD, Mitchener CD, Evans HE (eds) The biology of social insects. Westview, Press Boulder, pp 360–363
Mercer AR, Menzel R (1982) The effect of biogenic amines on conditioned and unconditioned responses to olfactory stimuli in the honey bee Apis mellifica. J Comp Physiol 145:363–368
Mercer AR, Mobbs PG, Davenport AP, Evans PD (1983) Biogenic amines in the brain of the honeybee Apis mellifica. l Tissue Res 234:655–677
Miller TA (1979) Nervous versus neurohormonal control of insect heartbeat. Am Zool 19:77–86
Myers PR (1974) Dopamine: Localization of uptake in the pedal ganglion of Quadrilla pustulosa (Pelecypoda). Tissue Cell 6:49–64
Nässel DR, Elekes K (1984) Ultrastructural demonstration of serotonin-immunoreactivity in the nervous system of an insect (Calliphora erythrocephala). Neursci Lett 48:203–210
Nässel DR, Elekes K (1985) Serotonergic terminal in the neuron sheath of the blowfly nervous system: electron microscopical immunocytochemistry and 5,7-dihydroxytryptamine labelling. Neuroscience 15:293–307
Nässel DR, Klemm N (1983) Serotonin-like immunoreactivity in the optic lobes of three insect species. Cell Tissue Res 232:129–140
Nässel DR, Laxmyr L (1983) Quantitative determination of biogenic amines and DOPA on the CNS of adult and larval blowflies Calliphora erythrocephala. Comp Biochem Physiol 75C:259–265
Nässel DR, Meyer EP, Klemm N (1985) Mapping and ultrastructure of serotonin-immunoreactive neurons in the optic lobes of three insect species. J Comp Neurol 232:190–204
Nishikawa N, Bennet GJ, Rua MA, Lu GW, Dubner R (1983) Immunocytochemical evidence for a serotoninergic innervation of dorsal column postsynaptic neurons in cat and monkey: Light and electron microscopic observations. Neuroscience 10:1333–1340
Panov AA (1964) Correlation in the ontogenetic development of the central nervous system in the house cricket Gryllus domesticus L. and the mole cricket Gryllotalpa gryllotalpa L. (Orthoptera, Gryllidea). Ent Rev 45:179–185
Pasik P, Pasik T, Pecci Saavedra J (1982) Immunocytochemical localization of serotonin at the ultrastructural level. J Histochem Cytochem 30:760–764
Pentreath VW, Berry MS (1975) Ultrastructure of the terminals of an identified dopamine-containing neurone marked by intracellular injections of radioactive dopamine. J Neurocytol 4:249–260
Pentreath VW, Berry MS (1978) Radioautographic study of 5-hydroxytryptamine nerve terminals in the central ganglia of Planorbis corneus: comparison with other species and characteristics of serotoninergic terminal. J Neurocytol 7:443–459
Robertson HA (1976) Octopamine, dopamine and noradrenaline content of the brain of the locust, Schistocerca gregaria. Experientia 32:552–553
Rude SR, Coggeshal RE, van Orden LS (1969) Chemical and ultrastructural identification of 5-hydroxytryptamine in an identified neuron. J Cell Biol 41:832–854
Sakaguchi DS, Murphey RK (1983) The equilibrium system of crickets: Physiology and morphology of an identified interneuron. Cell Tissue Res 216:79–96
Schürmann FW, Klemm N (1984) Serotonin-immunoreactive neurons in the brain of the honeybee. J Comp Neurol 225:570–580
Somogyi P, Takagi H (1982) A note on the use of picric acidparaformaldehyde-glutaraldehyde fixative for correlated light and electron microscopic immunocytochemistry. Neuroscience 7:1779–1783
Somogyi P, Hodgson AJ, Smith AD (1979) An approach to tracing neuron networks in the cerebral cortex and basal ganglia. Combination of Golgi-staining, retrograde transport of horseradish peroxidase and anterograde degeneration of synaptic bouton in the same material. Neuroscience 4:1805–1852
Spurr AR (1969) A low viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res 26:31–41
Sugawara R, Loher W (1986) Oviposition behaviour of the cricket Teleogryllus commodus: Observation of external and internal events. J Insect Physiol 32:179–188
Takeda S, Vieillemaringe J, Geffard M, Rémy C (19186) Imunohistological evidence of dopamine cells in the cephalic nervous system of the silkworm Bombyx mori. Co-existence of dopamine and α endorphin-like substance in neurosecretory cells of suboesophageal ganglion. Cell Tissue Res 243:125–128
Tyrer MN, Turner JD, Altman JS (1984) Identifiable neurons in the locust central nervous system that react with antibodies to serotonin. J Comp Neurol 227:313–330
Vieillemaringe J, Duris P, Geffard M, LeMoal M, Delaage M, Bensch C, Girardie J (1984) Immunohistochemical localization of dopamine in the brain of the insect Locusta migratoria migratorioides in comparison with catecholamine distribution determined by the histofluorescence technique. Cell Tissue Res 237:391–394
Wigglesworth VB (1957) The use of osmium in the fixation and staining of tissues. Proc R Soc Lond [Biol] 147:185–199
Author information
Authors and Affiliations
Additional information
Fellow of the Alexander von Humboldt-Stiftung
Rights and permissions
About this article
Cite this article
Elekes, K., Hustert, R. & Geffard, M. Serotonin-immunoreactive and dopamine-immunoreactive neurones in the terminal ganglion of the cricket, Acheta domestica: Light- and electron-microscopic immunocytochemistry. Cell Tissue Res. 250, 167–180 (1987). https://doi.org/10.1007/BF00214668
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00214668