Abstract
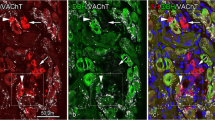
Substance P (SP) immunoreactivity in the guinea pig retina was studied by light and electron microscopy. The morphology and distribution of SP-immunoreactive neurons was defined by light microscopy. The SP-immunoreactive neurons formed one population of amacrine cells whose cell bodies were located in the proximal row of the inner nuclear layer. A single dendrite emerged from each soma and descended through the inner plexiform layer toward the ganglion cell layer. SP-immunoreactive processes ramified mainly in strata 4 and 5 of the inner plexiform layer. SP-immunoreactive amacrine cells were present at a higher density in the central region around the optic nerve head and at a lower density in the peripheral region of the retina. The synaptic connectivity of SP-immunoreactive amacrine cells was identified by electron microscopy. SP-labeled amacrine cell processes received synaptic inputs from other amacrine cell processes in all strata of the inner plexiform layer and from bipolar cell axon terminals in sublamina b of the same layer. The most frequent postsynaptic targets of SP-immunoreactive amacrine cells were the somata of ganglion cells and their dendrites in sublamina b of the inner plexiform layer. Amacrine cell processes were also postsynaptic to SP-immunoreactive neurons in this sublamina. No synaptic outputs onto the bipolar cells were observed.
Similar content being viewed by others
References
Brecha N, Hendrickson A, Floren I, Karten HJ (1982) Localization of substance P-like immunoreactivity within the monkey retina. Invest Ophthalmol Vis Sci 23:147–153
Brecha NC, Eldred W, Kuljis RO, Karten HJ (1984) Identification and localization of biologically active peptides in the vertebrate retina. In: Osborne N, Chader G (eds) Progress in retinal research. Pergamon Press, Oxford New York Toronto, pp 185–226
Brecha N, Cillufo M, Bolz J (1985) Identification of substance P immunoreactive ganglion cells in the rabbit retina. Soc Neurosci Abstr 11:1219
Brecha N, Johnson D, Bolz J, Sharma S, Parnavelas JG, Liebermann AR (1987) Substance P immunoreactive retinal, ganglion cells and their central axon terminals in the rabbit. Nature 327:155–158
Chang MM, Leeman SE (1970) Isolation of a sialogogic peptide from bovine hypothalamic tissue and its characterization as substance P. J Biol Chem 245:4784–4790
DeLanerolle NC, LaMotte CC (1982) The morphological relationships between substance P immunoreactive processes and ventral horn neurons in the human and monkey spinal cord. J Comp Neurol 207:305–313
Dick E, Miller RF (1981) Peptides influence retinal ganglion cells. Neurosci Lett 26:131–135
DiFiglia M, Aronin N, Leeman SE (1982) Light-microscopic and ultrastructural localization of immunoreactive substance P in the dorsal horn of monkey spinal cord. Neuroscience 7:1127–1139
Djamgoz MB, Dowling JE, Prince DJ (1983) Physiology of neuroactive peptides in the vertebrate retina. Biochem Soc Trans 11:686–689
Duner H, Euler US von, Pernow B (1954) Catecholamines and substance P in the mammalian eye. Acta Physiol Scand 31:113–118
Euler US von, Gaddum JH (1931) An unidentified depressor substance in certain tissue extracts. J Physiol (Lond) 72:74–87
Famiglietti EV, Kolb H (1975) A bistratified amacrine cell and synaptic circuitry in the inner plexiform layer of the retina. Brain Res 84:293–300
Famiglietti EV, Kaneko A, Tachibana M (1977) Neuronal architecture of on and off pathways to ganglion cells in carp retina. Science 198:1267–1269
Famiglietti EV, Brecha NC, Karten HJ (1980) Neural localization of substance P-like immunoreactivity in the rabbit retina. Soc Neurosci Abstr 6:212
Freed MA, Smith RG, Sterling P (1987) Rod bipolar array in the cat retina: pattern of input from rods and GABA-accumulating amacrine cells. J Comp Neurol 266:445–455
Glickman RD, Adolph AR, Dowling JE (1982) Inner plexiform circuits in the carp retina: effects of cholinergic agonists, GABA, and substance P on the ganglion cells. Brain Res 234:81–99
Kolb H (1979) The inner plexiform layer in the retina of the cat: electron microscopic observation. J Neurocytol. 8:295–329
Kolb H, Famiglietti EV (1976) Rod and cone pathways in the inner plexiform layer of cat retina. Science 186:47–49
Kolb H, Nelson R (1993) Off-alpha and off-beta ganglion cells in cat retina. II. Circuitry as revealed by electron microscopy of HRP stains. J Comp Neurol 329:85–110
Lee MY, Chun MH, Lee KH, Chung JW (1991) Two types of tyrosine hydroxylase immunoreactive neurons in the guinea pig retina. Invest Ophthalmol Vis Sci [Suppl] 32:1259
Léránth C, Fehér E (1983) Synaptology and sources of vasoactive intestinal polypeptide and substance P containing axons of the cat celiac ganglion. An experimental electron microscopic immunohistochemical study. Neuroscience 10:947–958
McGuire BA, Stevens J, Sterling P (1984) Microcircuitry of bipolar cells in cat retina. J Neurosci 4:2920–2938
McGuire BA, Stevens J, Sterling P (1986) Microcircuitry of beta ganglion cells in cat retina. J Neurosci 6:907–918
Mitrofanis J, Vigny A, Stone J (1988) Distribution of catecholaminergic cells in the retinal of the rat, guinea pig, cat, and rabbit: independence from ganglion cell distribution. J Comp Neurol 267:1–14
Oyster CW, Takahashi ES, Cilluffo M, Brecha NC (1985) Morphology and distribution of tyrosine hydroxylase-like immunoreactive neurons in the cat retina. Proc Natl Acad Sci USA 82:6335–6339
Pourcho RG, Goebel DJ (1988a) Substance P-like immunoreactive amacrine cells in the cat retina. J Comp Neurol 275:542–552
Pourcho RG, Goebel DJ (1988b) Colocalization of substance P and γ-aminobutyric acid in amacrine cells of the cat retina. Brain Res 447:164–168
Sagar SM, Weinstein L, Reinhard JF, Martin JB (1983) A comparison of the effects of kainic acid on somatostatin, substance P and dopamine in the rabbit retina. Brain Res 278:109–115
Stell WK, Ishida AT, Lightfoot DO (1977) Structural basis for on-and off-center responses in retinal bipolar cells. Science 198:1269–1271
Tornqvist K, Ehinger B (1988) Peptide immunoreactive neurons in the human retina. Invest Ophthalmol 29:680–686
Vacca LL, Hobbs J, Abrahams S, Naftchi E (1982) Ultrastructural localization of substance P immunoreactivity in the ventral horn of the rat spinal cord. Histochemistry 76:33–49
Vaney DI, Whitington GE, Young HM (1989) The morphology and topographic distribution of substance P-like immunoreactive amacrine cells in the cat retina. Proc R Soc Lond [Biol] 237:471–488
Voigt T, Wässle H (1987) Dopaminergic innervation of AII amacrine cells in mammalian retina. J Neurosci 7:4115–4128
Wulle I, Schnitzer J (1989) Distribution and morphology of tyrosine hydroxylase immunoreactive neurons in the developing mouse retina. Dev Brain Res 48:59–72
Yazulla S, Studholme KM, Zucker CL (1985) Synaptic organization of substance P-like immunoreactive amacrine cells in goldfish retina. J Comp Neurol 231:232–238
Zalutsky RA, Miller RF (1990) The physiology of substance P in the rabbit retina. J Neurosci 10:394–402
Zhang D, Yeh HH (1992) Substance P-like immunoreactive amacrine cells in the adult and the developing rat retina. Dev Brain Res 68:55–65
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, M.Y., Chun, M.H., Han, S.H. et al. Light- and electron-microscopic study of substance P-immunoreactive neurons in the guinea pig retina. Cell Tissue Res. 281, 261–271 (1995). https://doi.org/10.1007/BF00583395
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00583395