Summary
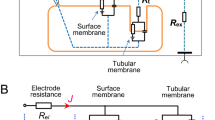
This is a study of the intracellular electrical potential, membrane resistance, and capacity of MDCK cells (epithelioid of renal origin) cultured in monolayers on a collagen couch. These monolayers have a transepithelial resistance of 256±12 (22) ohm cm2 (mean±standard error, and number of observations), and the cells have 61.6±6.3 (92) MΩ across their plasma membrane. The electrical capacity of the cells is 45.1±2.9 (63) pF and is much higher than expected for a cell of its size (diameter 14 μm, height 5 μm) and cannot be attributed to intercellular coupling, as no evidence of this type of connection was found in 20 pairs of neighboring cells. On the contrary, the high capacity is in keeping with previous studies using electron microscopy showing microvilli and a high degree of lateral infolding. The relationship between resistance and capacity was 1981±177 (61) Ω·μF. The cells have an intracellular potential of −40.5±15 (120) mV. Yet the shape of the distribution curve suggests that the actual value may be somewhat higher (some −50 mV). The current/voltage curve shows a marked asymmetry, and in some cells the voltage becomes time-dependent for large, depolarizing current pulses.
Similar content being viewed by others
References
Atkinson, M.M., Menko, A.S., Johnson, R.G., Sheppard, J.R., Sheridan, J.D. 1981. Rapid and reversible reduction in junctional permeability in cells infected with a temperature-sensitive mutant of avian sarcoma virus.J. Cell Biol. 91:573–578
Augustus, J., Bijman, J., Os, C.H. van, Slegers, J.F.G. 1977. High conductance in an epithelial membrane not due to extracellular shunting.Nature (London) 268:657–658
Cereijido, M., Stefani, E., Chávez de Ramirez, B. 1982. Occluding junction in theNecturus gallbladder.J. Membrane Biol. 70:15–25
Cereijido, M., Robbins, E.S., Dolan, W.J., Rotunno, C.A., Sabatini, D.D. 1978. Polarized monolayers formed by epithelial cells on a permeable and translucent support.J. Cell Biol. 77:853–880
Cereijido, M., Ehrenfeld, J., Meza, I., Martínez-Palomo, A. 1980. Structural and functional membrane polarity in cultured monolayers of MDCK cells.J. Membrane Biol. 52:147–159
Cereijido, M., Meza, I., Martínez-Palomo, A. 1981. Occluding junctions in cultured epithelial monolayers.Am. J. Physiol. 240:C96-C102
Cereijido, M., Rotunno, C.A. 1970. Introduction to the Study of Biological Membranes. Gordon and Breach, London
Cereijido, M., Rotunno, C.A., Robbins, E.S., Sabatini, D.D. 1978. Polarized epithelial membranes producedin vitro.In: Membrane Transport Processes. J.F. Hoffman, editor. Vol. 1, pp. 433–461. Raven Press, New York
Cereijido, M., Stefani, E., Martínez-Palomo, A. 1980. Occluding junctions in a cultured transporting epithelium: Structural and functional heterogeneity.J. Membrane Biol. 53:19–32
Flagg-Newton, J.L., Loewenstein, W.R. 1979. Experimental depression of junctional membrane permeability in mammalian cell culture. A study with tracer molecules in the 300 to 800 dalton range.J. Membrane Biol. 50:65–100
Furshpan, E.J., Potter, D.D. 1959. Transmission at the giant motor synapses of crayfish.J. Physiol. (London) 145:289–325
Herzlinger, D.A., Easton, T.G., Ojakian, G.K. 1982. The MDCK epithelial cell line expresses a cell surface antigen of the kidney distal tubule.J. Cell Biol. 93:269–277
Khuri, R.N., Agulian, S.K., Kalloghlian, A. 1972. Intracellular potassium in cells of the distal tubule.Pfluegers Arch. 335:297–308
Kuffler, S.W., Vaughan-Williams, E.M. 1953. Small-nerve junctional potentials. The distribution of small motor nerves to frog skeletal muscle, and the membrane characteristics of the fibres they inervate.J. Physiol. (London) 121:289–317
Ledbetter, M.L.S., Lubin, M. 1979. Transfer of potassium. A new measure of cell-cell coupling.J. Cell Biol. 80:150–165
Leighton, J., Brada, Z., Estes, L., Justh, G. 11969. Secretory activity and oncogenicity of a cell line (MDCK) derived from canine kidney.Science 163:472–473
Lever, J.E. 1979. Inducers of mammalian cell differentiation stimulate dome formation in a differentiated kidney epithelial cell line (MDCK).Proc. Natl. Acad. Sci. USA 76:1323–1327
Lever, J.E. 1981. Regulation of dome formation in kidney epithelial cell cultures.Ann. N.Y. Acad. Sci. 372:371–381
Loewenstein, W. 1981. Junctional intercellular communication: The cell-to-cell membrane channel.Physiol. Rev. 61:829–912
Loewenstein, W.R., Kanno, Y. 1964. Studies on an epithelial (gland) cell junction. I. Modifications of surface membrane permeability.J. Cell Biol. 22:565–586
Madin, S.H., Darby, N.B. 1958. As catalogued in: American Type Culture Collection Catalog of Strains.2:574–576
Malnic, G., Giebisch, G. 1972. Some electrical properties of distal tubular epithelium in the rat.Am. J. Physiol. 223:797–808
Martínez-Palomo, A., Meza, I., Beaty, G., Cereijido, M. 1980. Experimental modulation of occluding junctions in a cultured transport epithelium.J. Cell Biol. 87:736–745
Meza, I., Ibarra, G., Sabanero, M., Martínez-Palomo, A., Cereijido, M. 1980. Occluding junctions and cytoskeletal components in a cultured transporting epithelium.J. Cell Biol. 87:746–754
Misfeldt, D.S., Hamamoto, S.T., Pitelka, D.K. 1976. Transepithelial transport in cell culture.Proc. Natl. Acad. Sci. USA 73:1212–1216
Okada, Y., Sato, T., Inouye, A. 1975. Effects of potassium ions and sodium ions on membrane potential of epithelial cells in rat duodenum.Biochim. Biophys. Acta 413:104–115
Rabito, C.A., Tchao, R. 1980. [3H] ouabain binding during the monolayer organization and cell cycle in MDCK cells.Am. J. Physiol. 238:C43-C48
Rabito, C.A., Tchao, R., Valentich, J., Leighton, J. 1978. Distribution and characteristics of the occluding junctions in a monolayer of a cell line (MDCK) derived from canine kidney.J. Membrane Biol. 43:351–365
Rose, R.C., Schultz, S.G. 1971. Electrical potential profile across rabbit ileum.J. Gen. Physiol. 57:639–663
Simmons, N.L. 1981. Ion transport in tight epithelial monolayers of MDCK cells.J. Membrane Biol. 59:105–114
Valentich, J.D. 1981. Morphological similarities between the dog kidney cell line MDCK and the mammalian cortical collecting tubule.Ann. N.Y. Acad. Sci. 372:384–405
Weidmann, S. 1952. The electrical constants of Purkinje fibres.J. Physiol. (London) 118:348–360
Wiederholt, M., Agulian, S.K., Khuri, R.N. 1974. Intracellular potassium in the distal tubule of the adrenalectomized and aldosterone treated rat.Pfluegers Arch. 347:117–123
Windhager, E.E., Giebisch, G. 1965. Electrophysiology of the Nephron.Physiol. Rev. 45:214–244
Wondergem, R. 1981. Transmembrane potential of rat hepatocytes in primary monolayer culture.Am. J. Physiol. 241:C209-C214
Wright, F.S. 1971. Increasing magnitude of electrical potential along the renal distal tubule.Am. J. Physiol. 220:624–638
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Stefani, E., Cereijido, M. Electrical properties of cultured epithelioid cells (MDCK). J. Membrain Biol. 73, 177–184 (1983). https://doi.org/10.1007/BF01870440
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01870440