Abstract
Background. Tissue amyloid P component is a normal constituent of the human glomerular basement membrane (GBM) and is immunologically identical to the serum amyloid P component, a major DNA binding protein in serum. We postulate that DNA or nucleosome core particles could bind to human GBM via the amyloid P component.
Methods. An immunofluorescence study was used to detect the amyloid P component of the GBM. An enzyme-linked immunosorbent assay system was used to test the binding capacity of calf thymus DNA and chicken erythrocyte nucleosome core particles to a preparation of human GBM.
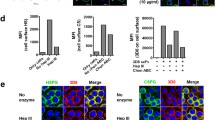
Results. Amyloid P component was detected along the capillary wall of the human glomerulus by immunofluorescence. DNA and nucleosome core particles bound to human GBM in a dose-dependent manner in the presence of Ca2+. Digestion of GBM with trypsin resulted in the reduction of binding of anti-serum amyloid P antibody, DNA, and nucleosome core particles to the GBM. Anti-serum amyloid P component (SAP) IgG blocked the binding of DNA and nucleosome core particles to the GBM.
Conclusion. DNA and nucleosome core particles bind to the GBM through amyloid P components.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: December 24, 1998 / Accepted: July 13, 1999
About this article
Cite this article
Morioka, T., Joh, K., Shimizu, F. et al. Nucleosome core particles and DNA bind to the human glomerular basement membrane (GBM): role of the amyloid P component of the GBM. Clin Exp Nephrol 4, 43–48 (2000). https://doi.org/10.1007/s101570050060
Issue Date:
DOI: https://doi.org/10.1007/s101570050060