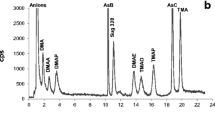
Abstract
The determination of arsenic species by the trapping of volatile hydrides prior to atomization in the light path of an atomic absorption spectrometer is described and its operation in the measurement of arsenic species in the marine environment are discussed. Examples are drawn from the analysis of Tamar estuary water and sediment interstitial (pore) waters and from studies of the temporal variation of dimethylarsenic in coastal waters. Improvements in both the design and operation of the technique have resulted in enhanced performance. Baseline resolution of inorganic arsenic, monomethylarsenic and dimethylarsenic is now possible and trimethylarsine is resolved. Ultraviolet photolysis of arsenobetaine and arsenocholine gives partial conversion to trimethylarsine oxide. This can be employed in the qualitative appraisal of the presence of trimethylarsenic species. Current detection limits (3 sigma) for inorganic, mono- and di-methylarsenic lie in the range 19 to 61 pg absolute, giving 19–61 ng/1 concentration detection limits for 1 ml samples. This can be improved even further by using larger sample volumes. The properties of the analysis system when presented with various arsenic species are described. A ca. 10% loss of arsenite occurs in samples stored at —20 °C and immediate freezing of samples in liquid nitrogen is recommended.
Similar content being viewed by others
References
A. G. Howard, M. H. Arbab-Zavar, S. Apte,Est. Coastal Shelf Sci. 1984,19, 493.
A. G. Howard, S. C. Apte, S. D. W. Comber, R. J. Morris,Est. Coastal Shelf Sci. 1988,27, 427.
J. S. Edmonds, K. A. Francesconi, J. R. Cannon, C. L. Raston, B. W. Skelton, A. H. White,Tel. Lett. 1977,18, 1543.
J. R. Cannon, J. S. Edmonds, K. A. Francesconi, C. L. Raston, J. B. Saunders, B. W. Skelton, A. H. White,Aust. J. Chem. 1981,34, 787.
J. F. Lawrence, P. Michalik, G. Tam, H. B. S. Conacher,J. Agric. Food Chem. 1986,34, 315.
H. Norin, A. Christakopoulos,Chemosphere 1982,11, 287.
H. Norin, R. Ryhage, A. Christakopoulos, M. Sandstroem,Chemosphere 1983,12, 299.
J. S. Edmonds, K. A. Francesconi,Nature 1981,289, 602.
K. Jin, T. Hayashi, Y. Shibata, M. Morita,Appl. Organomet. Chem. 1988,2, 365.
J. S. Edmonds, K. A. Francesconi, J. A. Hansen,Experientia 1982,38, 643.
J. S. Edmonds, K. A. Francesconi,J C S Perkin Trans. 1983,1, 2375.
J. S. Edmonds, K. A. Francesconi,Anal. Chem. 1976,48(13), 2019.
M. O. Andreae,Anal. Chem. 1977,49(6), 820.
A. G. Howard, M. H. Arbab-Zavar,Analyst 1981,106, 213.
A. M. M. De Bettencourt,Neth. J. Sea Res. 1988,22(3), 205.
W. R. Cullen, M. Dodd,Appl. Organomet. Chem. 1988,2, 1.
A. G. Howard, S. D. W. Comber,Appl. Organomet. Chem. 1989,3, 509.
S. D. W. Comber, A. G. Howard,Anal. Proc. 1989,26, 20.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Howard, A.G., Comber, S.D.W. Hydride-trapping techniques for the speciation of arsenic. Mikrochim Acta 109, 27–33 (1992). https://doi.org/10.1007/BF01243206
Issue Date:
DOI: https://doi.org/10.1007/BF01243206