Summary
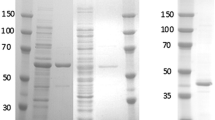
An NADH oxidase purified from the extreme thermophileThermus thermophilus HB8 is a monomeric flavoprotein with a 1 ∶ 1 ratio of flavin-adenine dinucleotide (FAD) to the polypeptide chain. It catalyzes in vitro the oxidation of reduced NADH or NADPH with the formation of H2O2. The gene encoding the NADH oxidase fromT. thermophilus HB8 was cloned, and its nucleotide sequence was determined. The molecular mass of 22,749 Da, as deduced from thenox gene, agrees with that of the purified NADH oxidase fromT. thermophilus HB8, as estimated by mass spectrometry. Thenox gene does not contain a GX4GK consensus sequence typical for nucleotide binding proteins. Thenox gene was overexpressed inEscherichia coli, and a protocol for the rapid purification of theE. coli-borneT. thermophilus NADH oxidase or its His6-tagged analogue was developed by using thermal denaturation step and affinity chromatography.
Similar content being viewed by others
References
Ahmed SA, Claiborne A (1989 a) The streptococcal flavoprotein NADH oxidase: evidence linking NADH oxidase and NADH peroxidase cysteinyl redox centers. J Biol Chem 264: 19856–19863
— — (1989 b) The streptococcal flavoprotein NADH oxidase: interactions of pyridine nucleotides with reduced and oxidized enzyme forms. J Biol Chem 264: 19864–19870
Boyer HW, Roulland-Doussix D (1969) A complementation analysis of the restriction and modification of DNA inEscherichia coli. J Mol Biol 41: 459–472
Cocco D, Rinaldi A, Savini I, Cooper JM, Bannister JV (1988) NADH oxidase from the extreme thermophileThermus aquaticus YT-1: purification and characterization. Eur J Biochem 174: 267–271
Dekker K, Yamagata H, Sakaguchi K, Udaka S (1991) Xylose (glucose) isomerase gene from the thermophileThermus thermophilus: cloning sequencing, and comparison with other thermostable xylose isomerases. J Bacteriol 173: 3078–3083
Dolin MI (1953) The oxidation and peroxidation of DPNH2 in extracts ofStreptococcus faecalis 10C1. Arch Biochem Biophys 46: 483–485
— (1955) The DPNH-oxidizing enzymes ofStreptococcus faecalis: II. The enzymes utilizing oxygen, cytochromec, peroxide and 2,6-dichlorophenol-indophenol or ferricyanide as oxidants. Arch Biochem Biophys 55: 415–435
Gottesman S, Halpern E, Trisler P (1981) Role of sulA and sulB in filamentation bylon mutants ofEscherichia coli K-12. J Bacteriol 148: 265–273
Hayashi M, Hasegawa K, Oguni Y, Unemoto T (1990) Characterization of FMN-dependent NADH-quinone reductase induced by menadione inEscherichia coli. Biochim Biophys Acta 1035: 230–237
Hohn B, Collins J (1980) A small cosmid for efficient cloning of large DNA fragments. Gene 11: 291–298
Hoskins DD, Whiteley HR, Mackler B (1962) The reduced diphosphopyridine nucleotide oxidase ofStreptococcus faecalis: purification and properties. J Biol Chem 237: 2647–2651
Jaiswal AK, Burnett P, Adesnik M, McBride OW (1990) Nucleotide and deduced amino acid sequence of a human cDNA (NQO-2) corresponding to a second member of the NAH(P)H: quinone oxidoreductase gene family: extensive polymorphism of the NQO-2 gene locus on chromosome 6. Biochemistry 29: 1899–1906
Kirino H, Oshima T (1991) Molecular cloning and nucleotide sequence of 3-isopropylmalate dehydrogenase gene (leuB) from an extreme thermophile,Thermus aquaticus YT-l. J Biochem 109: 852–857
Lauer G, Rudd EA, McKay DL, Ally A, Ally D, Backman KC (1991) Cloning, nucleotide sequence, and engineering expression ofThermus thermophilus DNA ligase, a homolog ofEscherichia coli DNA ligase. J Bacteriol 173: 5047–5053
Liu X-L, Scopes RK (1993) Cloning, sequencing and expression of the gene encoding NADH oxidase from the extreme anaerobic thermophileThermoanaerobium brockii. Biochim Biophys Acta 1174: 187–190
Ma Q, Wang R, Yang CS, Lu AYH (1990) Expression of mammalian DT-diaphorase inEscherichia coli: purification and characterization of the expressed protein. Arch Biochem Biophys 283: 311–317
Miller J (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Möller W, Amons R (1985) Phosphate-binding sequences in nucleotide-binding proteins. FEBS Lett 186: 1–7
Niimura Y, Ohnishi K, Yarita Y, Hidaka M, Masaki H, Uchimura T, Suzuki H, Kozaki M, Uozumi T (1993) A flavoprotein functional as NADH oxidase fromAmphibacillus xylanus EpOl: purification and characterization of the enzyme and structural analysis of its gene. J Bacteriol 175: 7945–7950
Park H-J, Kreutzer R (1994) Expression cloning of thenox, mdh, andldh genes fromThermus species encoding NADH oxidase, malate dehydrogenase and lactate dehydrogenase. Appl Microbiol Biotechnol 40: 676–681
— —, Reiser COA, Sprinzl M (1992 a) Molecular cloning and nucleotide sequence of the gene encoding a H2O2-forming NADH oxidase from the extreme thermophilicThermus thermophilus HB8 and its expression inEscherichia coli. Eur J Biochem 205: 875–879
—, Reiser COA, Kondruweit S, Erdmann H, Schmid RD, Sprinzl M (1992 b) Purification and characterization of a NADH oxidase from the thermophileThermus thermophilus HB8. Eur J Biochem 205: 881–885
Peter ME, Schimer NK, Reiser COA, Sprinzl M (1989) Mapping the effector region inThermus thermophilus elongation factor Tu. Biochemistry 29: 2876–2884
Ross PR, Claiborne A (1992) Molecular cloning and analysis of the gene encoding the NADH oxidase fromStreptococcus faecalis 10C1: comparison with NADH peroxidase and flavoprotein disulfide reductase. J Mol Biol 227: 658–671
Saeki Y, Nozaki M, Motsumoto K (1985) Purification and properties of NADH oxidase fromBacillus megaterium. J Biochem 98: 1433–1440
Schmidt HL, Stöcklein W, Danzer J, Kirch P, Limbach B (1986) Isolation and properties of an H2O-forming NADH oxidase fromStreptococcus faecalis. Eur J Biochem 156: 149–155
Schulze E, Benen JAE, Westphal AH, van Berkel WJW, de Kok A (1991) Binding studies of the dihydrolipoamide dehydrogenase component (E39) in the pyruvate dehydrogenase complex fromAzotobacter vinelandii. In: Curti B, Ronchi S, Zanetti G (eds) Flavins and flavoproteins 1990. W de Gruyter, Berlin, pp 569–572
Weisshaar M-P, Sprinzl M (1989) Cloning and expression of genes from the extreme thermophile bacteriumThermus thermophilus inE. coli. In: Schmid RD, Scheller F (eds) Biosensors — applications in medicine, environmental protection and process control. VCH, Weinheim, pp 417–419
Wierenga RK, de Maeyer MCH, Hol WGJ (1985) Interaction of pyrophosphate moieties with α-helices in dinucleotide binding proteins. Biochemistry 24: 1346–1357
Willem JH, van Berkel WJH, Snoek MC (1991) FAD-induced dimerization of apo-lipoamide dehydrogenase fromAzotobacter vinelandii andPseudomonas fluorescens. In: Curti B, Ronchi S, Zanetti G (eds) Flavins and flavoproteins 1990. W de Gruyter, Berlin, pp 585–588
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage vectors and host strains: nucleotide sequence of the M13mp18 and pUC19 vectors. Gene 33: 103–119
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Park, H.J., Erdmann, H. & Sprinzl, M. NADH oxidase ofThermus thermophilus HB8 overproduced fromEscherichia coli . Protoplasma 184, 104–110 (1995). https://doi.org/10.1007/BF01276906
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01276906