Abstract
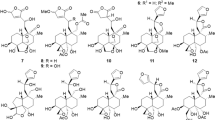
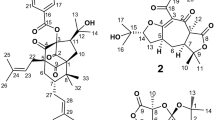
THE 16-acetyl derivative of digitalinum has been prefaced as follows. Digitalinum verum hexaacetate was obtained by the usual reaction of digitalinum verum with acetic anhydride in pyridine solution; this hexaacetate, using snail enzyme1, was partially deacetylated by potassium hydrogen carbonate to digitalinum verum diacetate, m.p. 181°–184°, (α)D–24.0° (CH3OH). The substance was separated by partition chromatography from the reaction mixtures, but it could not be induced to crystallize. The existence of acetoxyl group on carbon atom 16 of this glycoside has been confirmed by the facts that on treatment with alumina it affords a hydrogenated derivative, and that the molecular rotation difference between digitalinum verum and this compound is similar to the difference between gitoxigenin and oleandrigenin. Evidence is also given by the fact that it is hydrolysed into oleandrigenin on Mannich hydrolysis at low temperature (0°–5°).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Okano, A., Hoji, K., Miki, T., and Miyatake, D., Pharm. Bull., 5, 167 (1957).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
KANDA, Z., SEKIYA, A., KATAYAMA, K. et al. Chemistry and Pharmacology of 16-Acetyl Digitalinum Verum. Nature 184, 1414–1415 (1959). https://doi.org/10.1038/1841414b0
Issue Date:
DOI: https://doi.org/10.1038/1841414b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.