Abstract
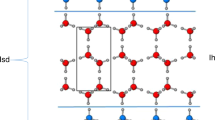
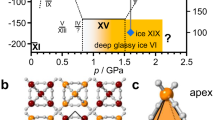
THE “inability of forming solid solutions with any compound” was attributed to water two years ago by Mironov and Bergman1, who refuted previous reports by Giguere and Maass2 and by Kubaschewski and Weber3 of the solubility of hydrogen peroxide in ice. Yet Kathleen Lonsdale4 directed attention to the fact that ice and ammonium fluoride are not only isomorphous but also give very similar diffuse star-shaped streaks on Laue X-ray patterns, which were attributed to thermal vibrations of hydrogen nuclei. This great similarity suggested that ice and ammonium fluoride might form mixed crystals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Proc. Acad. Sci. U.S.S.R., 81, 1081 (1951).
Can. J. Research, 18B, 66 (1940).
Z. Elektrochemie, 54, 200 (1950).
Nature, 158, 582 (1948).
J. Gen. Chem. (U.S.S.R.), 15, 724 (1945).
New Mexico School of Mines, Appendix H of Final Report on Thunderstorm Electricity (1951).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BRILL, R., ZAROMB, S. Mixed Crystals of Ice and Ammonium Fluoride. Nature 173, 316–317 (1954). https://doi.org/10.1038/173316a0
Issue Date:
DOI: https://doi.org/10.1038/173316a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.